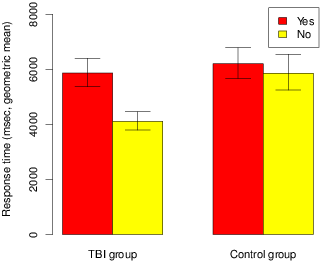
| Figure 1: Geometric mean response times (ms) for positive and negative answers to personal dilemmas (Control vs. TBI). Error bars are standard error, based on log transform. |
Judgment and Decision Making, vol. 7, no. 4, July 2012, pp. 478-487
Atypical moral judgment following traumatic brain injuryAna T. Martins.* Luis M. Faísca# Francisco Esteves% Angélica Muresan§ Alexandra Reis§ |
Previous research has shown an association between emotions, particularly social emotions, and moral judgments. Some studies suggested an association between blunted emotion and the utilitarian moral judgments observed in patients with prefrontal lesions. In order to investigate how prefrontal brain damage affects moral judgment, we asked a sample of 29 TBI patients (12 females and 17 males) and 41 healthy participants (16 females and 25 males) to judge 22 hypothetical dilemmas split into three different categories (non-moral, impersonal and personal moral). The TBI group presented a higher proportion of affirmative (utilitarian) responses for personal moral dilemmas when compared to controls, suggesting an atypical pattern of utilitarian judgements. We also found a negative association between the performance on recognition of social emotions and the proportion of affirmative responses on personal moral dilemmas. These results suggested that the preference for utilitarian responses in this type of dilemmas is accompanied by difficulties in social emotion recognition. Overall, our findings suggest that deontological moral judgments are associated with normal social emotion processing and that frontal lobe plays an important role in both emotion and moral judgment.
Keywords: moral judgment; social emotion recognition; frontal TBI.
For decades, moral psychology was dominated by Kantian-like rationalist theories that emphasized the role of conscious reasoning in the moral judgments of adults (Kohlberg, 1981; Piaget, 1932; for a recent review on moral reasoning, see Paxton & Greene, 2010). More recently, empirical researches suggested that moral judgment is predominantly intuitive, partly based on automatic emotional responses that are effortless and produced by unconscious processes. According to these perspectives, emotional processes play a crucial role in human decision making, including moral decisions (Greene & Haidt, 2002; Greene et al., 2001, 2004; Keltner, Horberg, & Oveis, 2006; Moll, Oliveira-Souza, & Eslinger, 2003; Slovácková, & Slovacek, 2007). The emotion based accounts of moral decision making draws support from a variety of empirical work, including behavioural and neuroimaging studies (Koenigs et al., 2007). For instance, several studies with clinical groups with brain focal lesions (see, for example, Anderson, Barrash, & Bechara, 2006) reveal an association between impaired emotional processing and disturbances in moral behaviour. Behavioural studies suggest that the manipulation of emotional states can influence the judgments in some moral situations (see, for example, Valdesolo & DeSteno, 2006; Wheatley & Haidt, 2005). In their neuroimaging study, Koenigs and colleagues (2007) show that moral decision making tasks activate the same brain areas involved in emotions processing.
The impairment of emotional behaviour has been commonly observed in patients with focal brain injuries (Greene et al., 2001; Greene, Turner, & Thompson, 2004; Milders et al., 2008; Moll et al., 2003; Prehn et al., 2007); specifically, patients with frontal lesions demonstrate a diminished emotional responsivity and an inadequate coping with anger and frustration in specific circumstances (Koenigs et al., 2007). This behavioural dysfunction appears to be related to a difficulty in processing specific social emotions (e.g., compassion, shame and guilt) that are closely linked to moral values (Adolphs, 2001; Adolphs, Baron-Cohen, & Tranel, 2002, Damasio et al., 2000; Eslinger, 1998). Despite these deficits regarding emotional regulation and response, frontal patients preserve their logical reasoning and declarative knowledge of social and moral norms (Burgess et al., 2006; Koenigs et al., 2007). In this sense, some frontal patients preserve intellectual abilities (for example, fund of general information, ability to repeat and reverse digits, mental arithmetic, verbal reasoning, nonverbal problem solving, verbal and visual anterograde memory, visuospacial perception, academic achievement) and maintain factual knowledge of social conventions and moral rules (Anderson et al. 2006). Interestingly, besides an inadequate emotional processing, an atypical pattern of moral judgements has also been observed in this clinic population (Eslinger, 1998; Gazzaniga, Ivry, & Mangun, 2009; Greene, & Haidt, 2002; Koenigs et al., 2007; Mendez, & Shapira, 2009; Moll et al., 2003).
The experimental study of moral judgement has frequently relied on the analysis of subject reactions to moral dilemmas, fictional social situations specifically designed to pit two alternative actions against each other; the choice will inevitably disfavour one of the parties involved in the dilemma (Greene & Haidt, 2002). Dilemmas are often subdivided into three categories: non-moral, impersonal moral and personal moral. A non-moral dilemma requires a decision that never involves a moral violation. In a moral dilemma the subject must typically choose between act and an omission, with the omission leading to greater harm. Green and colleagues (2004) distinguished between “personal” and “impersonal” moral violations. They consider a moral violation to be “personal” if it meets three criteria: “First, the violation must be likely to cause serious bodily harm. Second, this harm must befall a particular person or set of persons. Third, the harm must not result from the deflection of an existing threat onto a different party (Greene et al, 2004, p. 389).” These three criteria can be expressed in terms of “ME HURT YOU”. The “hurt” criterion picks out the most primitive kinds of harmful violations; “you” criterion ensures that the victim is vividly represented as an individual; and the “me” captures a notion of “agency”, requiring that the action springs in a direct way from the agent’s will, that it be “authored” rather than merely “edited” by the agent. A moral violation is impersonal if it fails to meet these three criteria.
Another important distinction between personal and impersonal moral dilemmas relates to their emotional level: personal moral dilemmas are characterized by high level of emotional intensity. Personal dilemmas are particularly intense because the moral violation option also represents an utilitarian response. From the utilitarian perspective, the morally correct course of action is the one that produces the greatest total benefit for all people affected (Choe & Min, 2011). In personal dilemmas, the utilitarian option involves a direct harmful action towards a specific individual that will favour the collective welfare; the internal conflict results from the strong emotional aversion to this utilitarian response and the consequent tendency to omit it. Securing the welfare of the collective by sacrificing a single person individual might be the ultimate moral goal of a society, however to choose this utilitarian option appears to be far from natural in normal individuals.
Brain damaged patients with impaired emotion processing often produce an abnormally utilitarian pattern of judgments on moral dilemmas. For example, patients with frontal lesions seem to choose to suffocate their own child in order to save a group of people who otherwise would die, including the baby (Greene & Haidt, 2002; Greene et al., 2004; Koenigs et al., 2007). Apparently, these patients often decide to secure the welfare of the collective even if it means sacrificing someone dear to them, an utilitarism attitude that suggest a diminished emotional responsivity. Anderson, Barrash, and Bechara (2006) argue that the abnormally high rate of utilitarian judgments observed in frontal brain injured patients with deficits in emotional response suggests that their decisions are mostly cognitive, intentional and conscious, uncontaminated by emotion. Functional neuroimaging studies of emotion recognition and moral judgment tasks highlight the pre-frontal cortex as dominant in the regulation of emotional behaviour as well as morality (Greene et al., 2004).
The frontal lesion patients reveal idiosyncratic response patterns to some dilemmas when compared to patients with lesions in other brain regions (Slovácková, & Slovacek, 2007). Several studies further investigating which regions in the frontal cortex would be dominant for judging moral actions led to little consensus among researchers. For example, Green and colleagues (2004) suggest that ventromedial (VMPFC), dorsolateral (DLPFC) and anterior cingulate (AC) cortex are involved in judging personal moral dilemmas, while Koenigs and colleagues (2007) postulate that the ventromedial (VMPFC) region is responsible not only for judgment of this type of dilemmas but also for processing more complex emotions (i.e., social emotions such as guilt or shame). Actually, both authors recognize the unique contributions of all three regions that together are relevant to moral judgments: VMPFC responsible for emotional responding; AC for cognitive conflict and DLPFC for the abstract reasoning.
Studies focusing on hemispheric dominance for emotion processing and moral judgment also provided little consensus. In order to understand whether the lateralization of the prefrontal cortex lesions leads to response differences in moral dilemmas with high emotional level, Tranel and colleagues (2002) and Young and colleagues (2006) examined subjects with unilateral and bilateral lesions. Both studies reported worse performance in patients with right and bilateral prefrontal cortex lesions. Following this line of research, Mendez and Shapira (2009) also concluded that subjects with right frontal cortex lesions showed an inadequate response pattern for emotion processing and abnormal judgment of personal moral dilemmas. These results seem to converge on the idea that frontal cortex has a neuronal circuit for “morality”, with predominance in the right hemisphere.
Regardless of which region in the prefrontal cortex is more dominant for moral judgments, evidence shows that damage to the prefrontal cortex impairs social and emotional behaviour, leading inevitably to some degree of social isolation. This pattern becomes more severe over time because social opportunities become scarcer and cognitive rehabilitation is nearly nonexistent (Bornhofen, & McDonald, 2008). Although several studies have examined emotional and behavioural changes in brain damaged patients, few studies pursued these changes specifically in frontal lesion subjects or investigated the possible association between social emotion processing and moral judgment. It is thus our first goal to compare the performance of frontal TBI patients with a group of healthy subjects on a moral judgment task. As a secondary objective, we ask whether TBI patients’ performance on the task differs when hemispheric lesion location (right, left, bilateral) and specific damaged prefrontal cortex region were considered. Since literature describes an association between adequate social emotion processing (e.g., compassion, shame, guilt) and judgment of moral dilemmas (Koenigs et al., 2007), our third aim is to analyze participants’ (TBI patients and controls) performance on a social emotion recognition ask and ask whether it correlates with utilitarian responses in moral dilemmas. This association may help us to understand the emotional and behavioural changes observed in subjects who suffered traumatic brain injury.
Table 1: Distribution of TBI patients according brain lesion localization.
Twenty-nine Traumatic Brain Injury (TBI) patients (17 male and 12 female) were assessed, with a mean age of 29.31 years (SD = 5.89) and mean number of years of education of 12.10 (SD = 3.25). On average, the post-traumatic period was 6.68 years (SD = 3.60). TBI patients were contacted in a private neurosurgical clinic in Algarve (n = 25) and at the Neurosurgical Department of Hospital of São José in Lisbon (n = 4) and invited to participate. The inclusion criteria were: (1) clinical diagnosis of frontal traumatic brain injury, (2) within 12 months or more, and (3) post-traumatic amnesia less than 24 hours. Exclusion criteria were: (1) previous clinical history of psychiatric disorder, (2) non-frontal traumatic brain injury, and (3) age over 40 years-old. From the 43 patients initially selected, 14 were excluded (six abandoned the experiment due to fatigue and physical indisposition, five did not show images of significant frontal brain damage and three were excluded due to technical failures during data collection). In order to get a more precise characterization, our TBI participants were submitted to a structural magnetic resonance imaging, being all of them tested with a Philips 1.5 Intera Scanner using the same image protocol.1 Subsequently, two independent judges (a neurosurgeon and a neuroradiologist) rated neuroimaging data for lesion location with the following grid criteria: frontal (anterior or posterior), frontal (orbitofrontal, medial, and dorsolateral), parietal (anterior or posterior), temporal (anterior or posterior) and occipital (anterior or posterior). Each lesion was characterized using the rules of abnormal focal signals (hypointense, hyperintense or mixed) (Table 1).
Forty-one healthy participants (25 male and 16 female) were recruited for this experiment and matched to TBI participants on age and years of education. Exclusion criteria included: (1) history of neurological disease; (2) self-reported depression and (3) history of traumatic brain injury. The mean age for the control participants was 27.98 years (SD = 5.73) and the mean number of years of education was 12.68 (SD = 3.66).
Participants were submitted to cognitive assessment through several tests: (1) Raven’s Progressive Matrices; (2) Wechsler Memory Scale; (3) Verbal Fluency Test (participants had to say as many name of animals as possible during 60 seconds); (4) Familiar Faces Recognition Test (naming task with three sub-tests: age, occupation and emotion perception of some familiar Portuguese’s faces); and (5) Trail Making Test—Part B. In addition, participant’s depression levels were evaluated using the Beck Depression Inventory (BDI) and TBI participants’ also respond to a Social and Emotional Maladjustment Scale (MSS), a seven items scale, specially developed for the present study (total score ranging from 7—absence of social maladjustment—to 49 —intense maladjustment; Cronbach’s alpha =.73).
The tests were applied in the same order and allow us to characterize participants’ cognitive functions and to evaluate potential confounding effects when evaluating group differences.
Table 2 reports means and standard deviation for cognitive results of both groups. The comparison between groups confirmed that they did not differ in any of the cognitive variables, except for Famous Faces Recognition Test, where the control group scored significantly higher (Cohen’s d indicating a large effect size). Furthermore, this difference was observed only when the TBI participants had to identify the emotional state of the famous face. There was also a statistically significant large difference between groups for depressive symptoms. TBI group presented higher scores for BDI, indicating more significant depressive symptoms compared to control group. On the Social and Emotional Maladjustment Scale, the TBI group showed relative high scores of social maladjustment (mean of 34.9, out of a maximum possible of 49 points).
Table 2: Sociodemographic, clinical and cognitive characterization of TBI and control groups.
M (SD)
M (SD)Note: BDI: Beck Depression Inventory (1: Inexistent or minimal Depression; 2: Minor depression; 3: Moderated Depression; and 4: Severe Depression); PMR: Raven’s Progressive Matrices (0–9: Very Inferior, I; 10–14: Inferior, II-; 15–24: Inferior, II; 25–49: Medium, III-; 50–74: Normal, III+; 75–84: Good, IV; 85–89: Good, IV+; 90–94: Very Good, V; 95–100: Very Good, V+); WMS: Wechsler Memory Scale (Minimal Quotient between 20 and 44 years old: 40; Maximum Quotient between 20 and 44 years old: 136); TMT: Trail Making Test—Part B (medium execution time: 60 seconds); VFT: Verbal Fluency Test (Total number of produced words); FFRT: Familiar Faces Recognition Test (4 points for each sub-test: age perception, occupation, and emotion. Total: 12 points); MSS: Social and Emotional Maladjustment Scale (minimum. 7: absence of social maladjustment to maximum.49: intense maladjustment); N/A: Not Applicable. *p ≤ .05; ** p ≤.01; ***p ≤.001 (significance obtained using Mann-Whitney non-parametric test).
In order to assess participants’ ability to recognize social emotions through facial expressions, a Go/No-Go Task was designed using Presentation 0.7 software (http://nbs.neurobs.com/presentation) to present stimuli and register answer accuracy and reaction times.
The stimuli used in this task was selected from Martins and Reis (2007) and consisted of black and white photographs with 44.46cm x 50cm depicting the face of an actor expressing one social emotion. Three actors were used to represent three social emotions (arrogance, jealousy and guilt) as well as a neutral expression. Pictures were selected after being evaluated by independent judges; only pictures with higher inter-rater agreement rates were used (Krippendorff’s alpha > .7).
The Go/No-Go task was organized in three blocks corresponding to each social emotion used in the study. Each block consisted of 24 stimuli: twelve (50%) represented the target emotion of the block (e.g., arrogance)—Go response—and twelve (50%) were distractors (four photos representative of neutral expression and eight photos representative of the other two social emotions)—No Go response. The three blocks presented sequentially always in the same order, separated by an interval of two minutes. Before each block and the next, participants were instructed which emotion they had to identify in the stimulus sequence. They were told to observe carefully each photograph presented in the sequence and, once they detect the target expression, they had to press a key as quickly as possible. Instructions were presented to participants during the training block, and were refreshed for each different experimental block. Each trial begins with a fixation cross, followed by one emotional stimulus presented for 500 ms. Then, a question mark (?) appeared for 1750 ms, the maximum time allowed to decide if the emotional stimulus presented corresponded to the block target emotion. Participants were instructed to press the key space whenever they identified the target emotion of each block. Once the response time limit expired, the next trial started automatically. Accuracy rates and reaction times were registered. In total, each participant responded to 72 trials; the score “1” was given for each correct answer (total score: minimum = 0; maximum = 36).
For the present study we adapted to Portuguese the 50 dilemmas originally created by Greene and colleagues (2004) and frequently used in the literature (see for example, Koenigs et al., 2007). The authors subdivided this original set of dilemmas in three categories: personal moral, impersonal moral and non-moral.2
In order to validate the classification imposed by the authors, an independent sample of ten judges rated the Portuguese version of each dilemma in terms of its emotional intensity using a seven-point scale (1—low emotional intensity; 7—high emotional intensity). The actions described in personal moral dilemmas were considered emotionally more intense than the actions described in impersonal moral dilemmas (mean ± sd: 5.71 ± 0.70 and M = 2.21 ± 0.47, respectively, computed across the dilemmas after averaging across subjects for each dilemma; Cohen’s d = 5.86; t = 3.25, p ≤ .001).
Twenty-two dilemmas were selected for the present study, six of which correspond to non-moral dilemmas, six to impersonal moral dilemmas and ten to personal moral dilemmas. Although participants respond to the three types of dilemmas, we only used moral personal dilemmas data.3 Stimuli were randomised in three different presentation orders. At the beginning of each trial, a cross (+) appears on the computer screen for 500 ms followed by the dilemma in text format (no time limit was imposed for reading the dilemma); when the participant finished reading the text, he will press a key in order to advance to the question they had to respond. This question was displayed in the screen for a maximum of 250 ms and the participant had to answer “yes” or “no”. The score “1” was assigned for each affirmative answer (“yes” responses always indicated commission of the proposed action). We use Presentation Software (version 0.7, http://nbs.neurobs.com/presentation) on a Samtron 73v computer for dilemmas presentation and for accuracy and response time registration.
All participants were Portuguese native speakers and were assessed in a quiet laboratory room of the University of Algarve. Two individual sessions were arranged for each participant, with mean time duration of 180 minutes. Informed consent was obtained from all subjects after the main objective of the study had been explained. At the beginning, participants filled in a demographic questionnaire, where they also had to provide neurological and medical information. Afterwards, the battery of cognitive tests described above was introduced. In the end, all participants performed the visual emotion recognition task and the moral judgment task.
We compute the proportion of affirmative answers given by subjects to the each personal dilemma. These dilemmas were constructed in such a way that an affirmative response (“yes”) corresponds to an utilitarian decision that implies damaging one of the parties (i.e., actors) of the story in order to maximize collective welfare. A prevalence of affirmative responses in this category of dilemmas would differ from the usual pattern found in healthy subjects that tended to respond negatively to these dilemmas (Koenigs, 2007). There is a systematic higher preference for affirmative responses in the TBI group compared to the control group (Table 3). This difference is significant in six of the ten personal dilemmas, and in the hypothesized direction for all 10.
Table 3: Proportion of affirmative answers to each personal dilemma.
Figure 1: Geometric mean response times (ms) for positive and negative answers to personal dilemmas (Control vs. TBI). Error bars are standard error, based on log transform. 
We converted the number of affirmative answers into a percentage and used it to evaluate subject’s tendency to give utilitarian responses. This composite measure is reliable (Cronbach’s alpha = .751).
There is a large significant difference between groups concerning the average proportion of affirmative answers in personal dilemmas (Cohen’s d = 1.24; t = 5.22, df = 68, p < .001): TBI patients show a higher proportion of affirmative answers (mean ± sd: 47.0% ± 23.3%) when compared to healthy participants (mean ± sd: 21.4% ± 17.8%).
Response times were analyzed separately for affirmative and negative answers using repeated measures analyse of variance model using the group (Controls vs. TBI) as a between-subject factor and type of answer (affirmative vs. negative) as within-subject factor. Response times were logarithmized before analyses to stabilised variance. The results showed a significant effect of type of answer [F (1, 60) = 14.34, p < .001] due to faster response times for negative answers. The advantage of negative answers seems to depend on the group [interaction group x type of answer: F (1, 60) = 7.58, p = .008]: TBI participants spent the same time deciding for yes and no responses (time difference = 32ms; Cohen’s d = 0.09; t = 0.71, df = 27, p = .483); however, no-responses were significantly faster than yes-responses in healthy group (time difference = 2112ms; Cohen’s d = 0.72; t = 4.79, df = 33, p < .001; see Figure 1). This interaction can be understood by assuming that the more frequent (hence more dominant) response is faster (Baron, Gürçay, Moore, & Starcke, K., in press): for the TBI group the two responses were about equally frequent, but for the healthy group the negative response was much more frequent.
As documented in the literature, there are differences in emotion recognition patterns according to hemispheric lateralization of the lesion (left, right or bilateral). Given this, we classified all participants according to the presence or absence of lesion into four subgroups: one subgroup exclusively with right frontal lesions (n = 11), one group with exclusively left frontal lesion (n = 10), a sub-group with bilateral lesion (n = 8) and a healthy sub-group (n = 41). The proportions of affirmative responses on personal dilemmas was analysed using a two-way ANOVA with right hemisphere lesion (present vs. absent) and left hemisphere (present vs. absent) as between-subject factors. The results showed a large significant effect of right-hemisphere [F (1, 66) = 13.86, p < .001]: participants with frontal right lesion (exclusively right frontal lesion and bilateral lesion) gave a higher percentage of affirmative responses compared to participants without right lesion (control participants and exclusively left frontal lesion participants) (mean percentages: 51.1% versus 24.9%, Cohen’s d = 1.24). The presence of left hemisphere lesion seems to have a medium but no significant effect on the percentage of affirmative responses to personal dilemmas [F (1, 60) = 1.0, p = .173; mean percentages: 43.2% versus 27.8%; Cohen’s d = 0.66].
The analysis of response times showed a significant effect of the type of answer [F (1, 58) = 4.5, p = .038], resulting from faster times for negative responses. Although the presence of any lesion increase response times, this effect is close to significant only for right hemisphere lesions [F (1, 58) = 3.9, p = .054]. However, the effect of left lesions seems to be similar for affirmative and negative answers [F (1, 58) = 2.2, p = .147].
In order to further investigate the association between lesion location and participants’ performance on judging personal moral dilemmas, we sub-divided the TBI group in three sub-groups according to the frontal cortex region affected: orbitofrontal (n = 8), medial (n = 3) and dorsolateral regions (n = 18). The nonparametric Kruskal-Wallis test identified a significant difference between groups regarding their performance on the task (H = 7.40, df = 2, p = .025). A post-hoc analysis using a Mann-Whitney test indicates that the difference results from a lower proportion of affirmative answers in the dorsolateral group compared to the other two groups (p < .05).
Given the close relationship described in the literature between moral judgment and emotional processing, we looked for the existence of an association between the proportion of affirmative responses on personal dilemmas and the performance on a visual social emotion recognition task. The results suggest a negative association between these two tasks (Pearson r = −.374, p = .003), indicating that, when the answer to the personal moral dilemmas is more utilitarian, performance on the social emotion recognition task is worse. We also found a reduction in social emotion recognition in the TBI group (r = −.607, t(58) = −5.82, p < .001).4
The pattern of impaired moral and social behaviour consistently observed in certain brain injured subjects stimulated, over the last decades, a significantly increasing amount of research studies oriented not only to identifying cerebral correlates for social emotion processing and moral judgment but also to see how these two processes are related. Some of these studies (for example, Greene et al., 2004, and Koenigs et al., 2007) found an atypical social and moral behavioural pattern in subjects with frontal brain lesions and suggested a possible association between this pattern and impaired social emotion processing and moral judgment. Although there is some work developed in this field, few studies were dedicated to the performance of TBI patients on social emotion recognition and moral judgment and even less to the association between these two tasks in these patients.
We examined how frontal TBI subjects performed on a set of highly emotional moral dilemmas and compared their results with those of a matched group of healthy participants. The results showed that the TBI group had a higher proportion of affirmative responses in personal moral dilemmas compared to controls. These results are similar to those obtained by other authors (Greene et al., 2001, 2004; Koenigs et al., 2007), and are explained by the nature of the particular type of dilemma used. When confronted with a moral dilemma that pit compelling considerations of aggregate welfare against highly emotionally aversive behaviour, frontal TBI patients decide to secure the welfare of the collective even if it means sacrificing someone. This response pattern suggests a diminished responsivity to the emotional load of the dilemma, allowing them to respond to utilitarian considerations without the emotional conflict that leads most healthy subjects to respond negatively to these dilemmas.
The analysis of response times supports the previous interpretation: although TBI patients showed longer response times than controls, there was no time difference between affirmative and negative responses. But controls were clearly faster when they answered negatively to personal dilemmas than when they gave an affirmative response. The longer times in affirmative/utilitarian responses given by healthy subjects suggest sensitivity to the high emotional load of the dilemma not observed in the brain damaged group, which resulted in conflict.
Ciaramelli and colleagues (2007), as well as Koenigs and colleagues (2007), proposed that proper social emotions processing is an indispensable prerequisite for normal resolution of moral actions, suggesting a common neuronal substrate for emotion processing and moral judgment. Moreover, impaired comprehension of social emotions in others (i.e., reduced ability to empathize), which is frequently observed in frontal brain damaged patients, severely compromises the ability to adequately judge moral dilemmas with high emotional intensity. Thus, subjects with frontal brain lesions tend to use only one utilitarian pattern when solving moral dilemmas, apparently revealing lack of moral conflict. Due to their impaired emotional processing, our TBI patients tend to assess personal moral dilemmas based solely on cognitive criterion, perhaps engaging in conscious abstract reasoning processes and cost-benefit analysis. In the control group, the sensitivity to dilemmas’ emotional load may have contributed to an intuitive and automatic emotional assessment of the dilemma that directs them to a fast negative response.
Based on our results, and similarly to Shamay-Tsoory and Aharon-Peretz (2007), we suggest the prefrontal cortex as an important regulator for moral judgment.
Similarly to other studies (see for example, Tamietto et al., 2007), we also asked whether moral dilemmas assessment depended on the lateralization of the lesion in the prefrontal cortex. We found that the existence of a right-hemisphere lesion is significantly associated to a larger preference for utilitarian decision in personal moral dilemmas, as well as for longer response times. The presence of a left-hemisphere lesion may also have a positive medium contribution for utilitarian decisions, but its effect was not significant in our study. In spite of inter-hemispheric cooperation theory both for social emotions processing (Braun et al., 2005; Tamietto et al., 2007) and for moral judgment, we might suggest, following Demaree and colleagues (2005), that the right hemisphere may play a more decisive role than the left in moral judgments (at least their emotional aspect).
Concerning the role of specific prefrontal cortex regions in moral judgment, Greene and colleagues (2004) and Koenigs and colleagues (2007) highlight the importance of the ventromedial cortex. When we divided our TBI group in orbitofrontal, ventromedial and dorsolateral subgroups, according to the localisation of the lesion, we verified that the preference for utilitarian responses was smaller in the dorsolateral subgroup. According to Ciaramelli and colleagues (2007) the ventromedial cortex is critical to take a firm stand against personal moral violations, perhaps by mediating or self-focused emotional reactions that affect moral decisions.
We also sought to understand whether there was an association between the response pattern on moral dilemmas and performance on the visual recognition of social emotions (arrogance, jealousy and guilt). The results showed a negative association between the social emotions recognition and the proportion of utilitarian responses to personal moral dilemmas. These findings suggest that an atypical preference for utilitarian responses is associated with deficits in social emotion recognition, although recognition of emotion in faces is only part of the general emotional sensitivity that may be relevant.
Although our TBI patients revealed more difficulties than the controls on moral judgment and social emotion recognition tasks, and in spite of the association between these two tasks for them, our results did not show any difficulties on cognitive tests. These results converge with those found by Burgess and colleagues (2006) who propose that subjects with frontal brain lesions are able to frequently accomplish high complex everyday tasks. Furthermore, these results suggest that the evaluation of emotional moral and non-emotional stimuli depend on different and independent neuronal circuits.
Because it is described in the literature that TBI patients often display depressive symptoms, which could bias our interpretation of the data from moral judgment and social emotional recognition tasks, we addressed this potential problem through Beck Depression Inventory (BDI) and observed values representing only mild depression in the clinical group. According to Eslinger, Parkinson and Shamay (2002), some depression is expected in aware TBI patients because of their gradual disengagement from the society. Depressive symptoms did not seem to mediate the effect of TBI, however. Although the correlation between BDI and affirmative responses to personal dilemmas was substantial (r = .464, p < .001), a regression of the dilemma score on TBI and BDI found no significant effect of BDI, although the TBI coefficient was significant (p = .003).5 Thus, it would seem that the correlation of BDI with moral judgment is more likely the result of its correlation with TBI, rather than the cause of it.
In conclusion, we believe that our results are consistent with the current model that supports joint action between the intuitive/emotional and the conscious/rational systems in moral judgment (Koenigs et al., 2007). Further investigation is necessary in order to obtain a more detailed characterization of the emotional system and its neural correlates.
Adolphs, R. (2001). The neurobiology of social cognition. Current Opinion in Neurobiology, 77, 231–239.
Adolphs, R., Baron-Cohen, S., & Tranel, D. (2002). Impaired recognition of social emotions following amygdala damage. Journal of Cognitive Neuroscience, 14, 1264–1274.
Anderson, S. W., Barrash, J., & Bechara, A. (2006). Impairments of emotion and real-world complex behavior following childhood- or adult-onset damage to ventromedial prefrontal cortex. Journal of the International Neuropsychological Society, 12, 224–235.
Baron, J., Gürçay, B., Moore, A. B., & Starcke, K. (in press). Use of a Rasch model to predict response times to utilitarian moral dilemmas. Synthese (special issue on psychological models of (ir)rationality and decision making, edited by C. Witteman & W. van der Hoek). DOI 10.1007/s11229-012-0121-z
Bornhofen, C., & McDonald, S. (2008). Emotion perception deficits following traumatic brain injury: A review of the evidence and rationale for intervention. Journal of the International Neuropsychological Society, 14, 511–525.
Braun, M., Traue, H., Frisch, S., Deighton, R., & Kessler, H. (2005). Emotion recognition in stroke patients with left and right hemispheric lesion: Results with a new instrument-the FEEL Test. Brain and Cognition, 58(2), 193–201.
Burgess, P. W., Alderman, N., Forbes, C. (2006). The case for the development and use of “ecologically valid” measures of executive functions in experimental and clinical neuropsychology. Journal of the International Neuropsychological Society, 12, 194–209.
Choe, S. Y. & Min, K. H. (2011). Who makes utilitarian judgments? The influences of emotions on utilitarian judgments. Judgment and Decision Making, 6(7), 580–592.
Ciaramelli, E., Muccioli, M., Làdavan, E., & Pellegrino, G. (2007). Selective deficit in personal moral Judgment following ventromedial pre-cortex. Social Cognitive and Affective Neuroscience, 17, 344–354.
Damásio, A. R., Grabowski, T. J., Bechara, A., Damásio, H., Ponto, L. L. B., Parvizi, J., & Hichwa, R. D. (2000). Subcortical and cortical brain activity during the feeling of self-generated emotions. Nature Neuroscience, 3, 1049–1056.
Demaree, H., Everhart, D., Youngstorm, E., & Harrison D. (2005). Brain lateralization of emotional processing: Historical roots and a future incorporating dominance. Behavioral and Cognitive Neuroscience Reviews, 4, 3–20.
Eslinger, P.(1998). Neurological and neuropsychological bases of empathy. European Neurology, 39, 193–199.
Eslinger, P., Parkinson, K., & Shamay, S. G. (2002). Empathy and social-emotional factors in recovery from stroke. Current opinion in neurology, 15, 91–97.
Gazzaniga, M., Ivry, R., & Mangun, G. (2009). Cognitive Neuroscience: the biology of the mind, 3 ed. International Student Edition: New York, London.
Green, R., Turner, G., & Thompson, W. (2004). Deficits in facial emotion perception in adults with recent traumatic brain injury. Neuropsychologia, 42, 133–141.
Greene, J., & Haidt, J. (2002). How (and where) does moral judgement work? Trends in Cognitive Sciences, 6(12), 517–523.
Greene, J., Nystrom, L., Engell, A., Darley, J., & Cohen, J. (2004). The neural bases of cognitive conflict and control in moral judgment. Neuron, 44, 389–400.
Greene, J., Sommerville, R., Nystrom, L., Darley, J., & Cohen, J. (2001). An fMRI investigation of emotional engagement in moral judgment. Science, 293, 2105–2108.
Kahane, G., & Shackel, N. (2010). Methodological issues in the neuroscience of moral judgment. Mind and Language, 25, 561–582.
Keltner, D., Horberg, E., & Oveis, C. (2006). Emotional intuitions and moral play. Social Justice Research, 19, 208–217.
Koenigs, M., Young, L., Adolphs, R., Tranel, D., Cushman, F., Hauser, M., & Damásio, A. (2007). Damage to the prefrontal cortex increases utilitarian moral judgements. Nature, 446, 908–911.
Kohlberg, L. (1981). Essays on moral development, Vol 1. San Francisco: Harper & Row.
Martins, A., & Reis, A. (2007). Validação de estímulos para construção de paradigmas para o estudo do reconhecimento de emoções. Provas de Aptidão Pedagógica e Capacidade Científica, Universidade do Algarve.
Mendez, M. F., & Shapira, J. S. (2009). Altered emotional morality in frontotemporal dementia. Cognitive Neuropsychiatry, 14, 165–179.
Milders, M., Ietswaart, M., Crawford, J., & Currie, D. (2008). Social behaviour following traumatic brain injury and its association with emotion recognition, understanding of intention, and cognitive flexibility. Journal of the International Neuropsychological Society, 14, 318–326.
Moll, J., Oliveira-Souza, P., & Eslinger, P. (2003). Morals and the human brain: a working model. Neuroreport, 14, 299–305.
Paxton, J. M. & Greene, J. D. (2010). Moral reasoning: Hints and allegations. Topics in Cognitive Science, 2, 511–527.
Piaget, J. (1965). The moral judgment of the child. New York: Free Press. (Original work published 1932).
Prehn, K., Wartenburger, I., Mériau, K., Scheibe, C., Goodenough, O. R., Villringer, A., Meer, E., & Heekeren, H. R. (2007). Individual differences in moral judgment competence influence neural correlates of socio-normative judgments. Social Cognitive and Affective Neuroscience, 3, 33–46.
Raven, J., Raven, J. C., & Court, J. H. (1998a). Manual for Raven’s Progressive Matrices and Vocabulary Scales. Section 1: General overview. Oxford, England: Oxford Psychologists Press/San Antonio, TX: The Psychological Corporation.
Reitan, R. M. (1958). Validity of the Trail Making test as an indicator of organic brain damage. Perceptual Motor Skills, 8, 271–276.
Shamay-Tsoory, S. G, & Aharon-Peretz, J. (2007). Dissociable prefrontal networks for cognitive and affective theory of mind: A lesion study. Neuropsychologia, 45, 3054–3067.
Slovácková, B., & Slovácek, L. (2007). Moral judgement competence and moral attitudes of medical students. Nursing Ethics, 14(3), 320–328.
Tamietto, M., Adenzato, M., Geminiani, G., & Gelder, B. (2007). Fast recognition of social emotions takes the whole brain: Interhemispheric cooperation in the absence of cerebral asymmetry. Neuropsychologia, 4, 836–843.
Tranel, D., Bechara, A., & Denburg, N. L. (2002). Asymmetric functional roles of right and left ventromedial prefrontal cortices in social conduct, decision-making, and emotional processing. Cortex,38, 589–612.
Valdesolo, P., & DeSteno, D. (2006). Manipulations of emotional context shape moral judgment. Psychological Science, 17, 476–477.
Wechsler, D. (1945). A standartized memory scale for clinical use. Journal of Psychology, 19, 87–95.
Wheatley, T., & Haidt, J. (2005). Hypnotic disgust makes moral judgments more severe. Psychological Science, 16, 780–784.
Young, L., Cushman, F., Adolphs, R., Tranel, D., & Hauser, M. (2006). Does emotion mediate the effect of an action’s moral status on its intentional status? Neuropsychological evidence. Journal of Cognition and Culture, 6, 265–278.
This document was translated from LATEX by HEVEA.